Alzheimer’s Disease Treatments: Money Rules
By John McDougall, MD
A larger trial found the drug, lecanemab, sold under the brand name Leqembi, slowed the progression of Alzheimer’s disease by a meager 27 percent, at a price of $26,500 for one year. Swelling and bleeding in the brain occurred in about 20 percent of patients vs. 9 percent in the placebo group in the 18-month Clarity trial.
A year before, in 2021, another similar, FDA-approved drug, Aduhelm, at a cost of $56,000 annually, showed such poor benefits that it was criticized by scientists and physicians worldwide. Neither of these drugs is intended to be a cure.
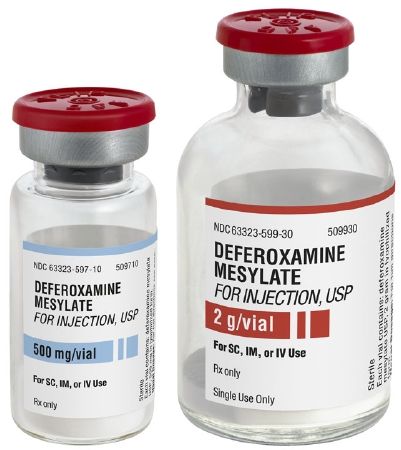
An inexpensive and safe generic treatment for this common form of dementia was tested in the 1980s. A two-year randomized trial of 63 patients performed with deferoxamine, a medication made by bacteria, found that the performance of the skills of daily living was 2 times better with the treatment. Alzheimer’s Disease is due to aluminum poisoning. This drug removes the toxic metal aluminum from the body and brain and therefore would be acting as a cure. One minor limitation was due to the twice-daily intramuscular injections. However, the truth is it never became part of medical care because deferoxamine is not patentable and could never be a runaway blockbuster for pharmaceutical companies.
See: Lancet. 1991 Jun 1;337(8753):1304-8; Ther Drug Monit. 1993 Dec;15(6):602-7; J Alzheimers Dis 40 (2014) 765–838
Learn more about how to preserve and improve cognitive decline along with effective treatments for Alzheimer’s disease in our Aging Gracefully mini-course.
If you need medical support, see if the 12-Day McDougall Program is right for you.
Recommended Articles

When Friends Ask: Why Do You Avoid Adding Vegetable Oils?

Human Genes are Turned On and Off by Diet





Join the Conversation!
Have a question, a helpful tip, or a story from your journey? Add it below - your voice matters.